A new method of tissue expansion technology may help increase the ability to observe and label proteins involved in synaptic connections. Researchers from the Massachusetts Institute of Technology say their findings improve neural imaging capabilities for scientists.
Tissue expansion technologies have long been an essential tool in observing cellular structures too small for a traditional microscope, particularly viewing those proteins involved in cellular connections. But prior methods of tissue expansion technologies often resulted in chemical bonds forming in the exact location that fluorescent antibody labels needed to attach to proteins.
This makes labeling many proteins involved in neural connections, or synapses, infeasible.
Now, with the expansion of technology first developed by co-author Dr. Kwanghun Chang in 2016, the binding sites for those fluorescent antibody label markers are much less likely to be blocked by a chemical bond. The development of epitope-preserving magnified analysis of proteome (eMAP) has proven particularly useful in identifying and labeling protein connections in synapses.
Tissue expansion technologies involve the injection of an acrylamide mesh into tissue, which attaches itself to all proteins within the tissue. Chemical bonds would then be formed between the mesh and tissue, providing a secure attachment that allowed the proteins to stay in place when expanded.
Traditional tissue expansion methods provided a detailed image of cellular structure otherwise unseen. Those very same chemical bonds that enabled the tissue to accurately expand, however, often blocked researchers from labeling, and identifying, a number of proteins.
With eMAP technology, the injected mesh no longer forms chemical bonds, but fully enmeshes itself in the tissue, leaving previously blocked binding sites open to protein label markers. This has allowed researchers to increase the number of synaptic proteins identified with antibody labels by 27%.
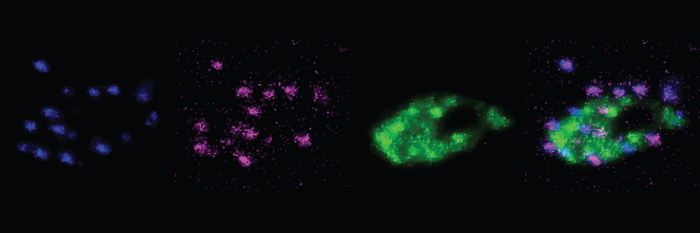
“eMAP preserves the fine-scale molecular architecture of synapses and can facilitate high-throughput analysis of macromolecular assemblies with its exceptional compatibility with the large library of off-the-shelf antibodies,” the team writes.
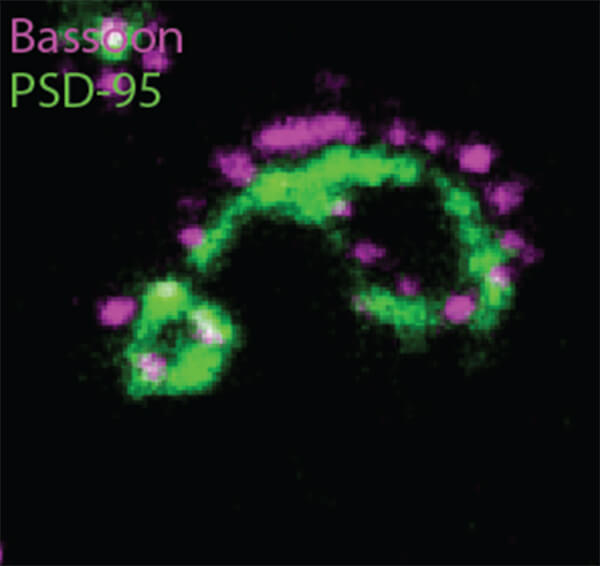
This new method provided 14 new fluorescent antibody labels to attach to previously unlabeled proteins, such as Bassoon, Piccolo, and Homer1. The multiscale imaging involved with eMAP also produces a far more vivid picture of synapses than previous methods, both individually and along dendrites, or neuronal connections.
The images produced through eMAP not only allowed a number of new proteins to be labeled in a synapse, but the precise location and arrangement of the proteins, as well. And since this technology is far less labor intensive than previous methods of synaptic imaging, it can be applied multiple times to the same tissue, greatly increasing the number of antibody labels attached to proteins in a single synapse.
This new technology may help further expand researchers’ knowledge and understanding of synaptic connections, and provide crucial insight into the function of synaptic proteomes moving forward.
The paper is published in Science Advances.
Article written by Adam Swierk