Recently developed technology may offer insight on brain development and repair. The unique neural interfaces integrate cutting-edge 3D bioelectronic devices with extremely developed 3D human brain cells. The objective is to provide accurate in vitro investigations of how human neural pathways grow and heal themselves.
Researchers hope this technology will give solutions for brain healing in the aftermath of neurotrauma and neurodegenerative disorders. The neural interfaces were created by Northwestern University researchers and scientists at the University of Illinois – Chicago (UIC) and Shirley Ryan AbilityLab.
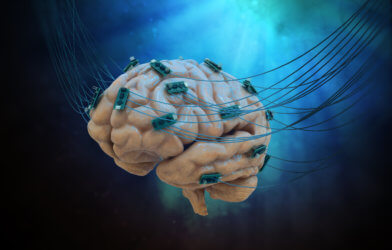
The cortical microspheres, or “mini-brains,” utilized in the investigation were generated from living human pluripotent stem cells. Scientists were able to construct a “miniature lab in a dish” particularly suited to observe the mini-brains and gather multiple kinds of data at the same time by utilizing a 3D brain interfacing device created by the group. Electrodes were used to measure electrical activity.
Researchers inserted small heating components to either maintain a steady temperature or, in some circumstances, to add stress by deliberately overheating the cultures. They also used microscopic probes like oxygen sensors and miniature LED lights to conduct optogenetic studies. For example, scientists inserted genes into the cells that enabled them to regulate brain function according to the color of light flashed upon them.
This system allowed scientists to conduct sophisticated research on human tissue without engaging individuals directly or undertaking intrusive tests. Anyone, in principle, may give a limited quantity of their cells (e.g., blood sample, skin biopsy), to which scientists can modify to create a small brain spheroid with the same genetic makeup as the person. Researchers think they will be able to develop better, innovative therapies faster by integrating this technology with a customized medicinal strategy based on human stem cell-derived neural cells.

“The advances spurred by this research will offer a new frontier in the way we study and understand the brain,” says Dr. Colin Franz from Shirley Ryan AbilityLab and co-lead author on the paper who led the testing of the cortical spheroids. “Now that the 3-D platform has been developed and validated, we will be able to perform more targeted studies on our patients recovering from neurological injury or battling a neurodegenerative disease.”
“This is just the beginning of an entirely new class of miniaturized, 3-D bioelectronic systems that we can construct to expand the capacity of the regenerative medicine field. For example, our next generation of devices will support the formation of even more complex neural circuits from the brain to muscle, and increasingly dynamic tissues like a beating heart,” adds Yoonseok Park, a postdoctoral fellow at Northwestern University and co-lead author.
Existing electrode arrays for cell culturing are incapable of replicating intricate natural structures like those that make up the human brain because they are two-dimensional and flat. Furthermore, even in a 3-D system, integrating multiple components into a tiny 3-D structure is exceedingly difficult. However, with this advancement, a whole new class of 3-D bioelectronics devices has been developed for use in regenerative medicine.
“Now, with our small, soft 3-D electronics, the capacity to build devices that mimic the complex biological shapes found in the human body is finally possible, providing a much more holistic understanding of a culture,” said Northwestern’s John Rogers, who led the technology development using technology similar to that found in phones and computers. “We no longer have to compromise function to achieve the optimal form for interfacing with our biology.”
The technology will be used to further investigate neurological disorders, test medicines and treatments with clinical potential, and evaluate various cell models acquired from patients. This insight will therefore allow for a greater understanding of individual variations, which may explain the broad range of results found in neurological rehabilitation.
“As scientists, our goal is to make laboratory research as clinically relevant as possible,” said Kristen Cotton, research assistant in Dr. Franz’s lab. “This 3-D platform opens the door to new experiments, discovery, and scientific advances in regenerative neurorehabilitation medicine that have never been possible.”
This study was published in the March 2021 issue of Science Advances.








-392x250.jpg)