Deep Brain Stimulation (DBS) enables scientists and physicians to deliver electrical impulses via electrodes to the brain area that regulates motor control. Several studies have revealed it to be a tried-and-true method for controlling undesired muscle activity, however symptoms persist as soon as the electrodes stop firing. For individuals to ease their symptoms, they must undergo DBS repeatedly.
According to Aryn Gittis, an associate Biology professor at the Carnegie Mellon College of Science and a faculty member of the Neuroscience Institute, a more targeted approach could solve the issue – and she and her team were the right people for the job.
“By finding a way to intervene that has long-lasting effects, our hope is to greatly reduce stimulation time, therefore minimizing side effects and prolonging battery life of implants,” said Gittis, who paved the way for this treatment strategy.
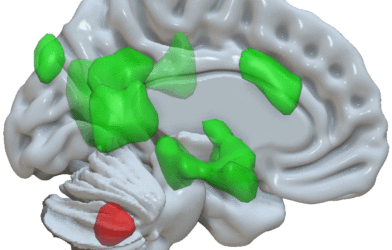
In 2017, she and her team identified particular brain cells in the motor cortex that are capable of being directly manipulated. Stimulating these neurons directly, enabled individuals with Parkinson’s disease to have long-term alleviation of their uncontrollable muscle spasms.
Optogenetics, a method that harnesses light to regulate brain cells that have been genetically manipulated, was the primary technique of that study. However, this method cannot presently be applied to people. Therefore, Gittis and her team have been working to develop a technique that may be more easily applied to individuals living with Parkinson’s. Instead of the usual constant stimulation method, according to her research, smaller, more precise doses of electrical impulses used on mice showed greater success.
“This is a big advance over other existing treatments,” Gittis said. “In other DBS protocols, as soon as you turn the stimulation off, the symptoms come back. This seems to provide longer lasting benefits — at least four times longer than conventional DBS.”
This novel technique only requires brief pulses of electrical activity to reach specific groups of neurons in the region of the brain called the globus pallidus. This motor cortex region is located in a smaller area called the basal ganglia. According to Gittis, for years specialists have tried developing methods of delivering stimulation in a way that allows only certain cell groups to be affected. “That concept is not new. We used a ‘bottom up’ approach to drive cell type specificity. We studied the biology of these cells and identified the inputs that drive them. We found a sweet spot that allowed us to utilize the underlying biology,” she said.
Despite several compelling hypotheses, according to Teresa Spix, the paper’s lead author, experts still do not completely comprehend the method behind DBS. “We’re sort of playing with the black box. We don’t yet understand every single piece of what’s going on in there, but our short burst approach seems to provide greater symptom relief. The change in pattern lets us differentially affect the cell types,” said Spix.
She is thrilled by the significant relation between her research and clinical investigations. “A lot of times those of us that work in basic science research labs don’t necessarily have a lot of contact with actual patients. This research started with very basic circuitry questions but led to something that could help patients in the near future,” Spix said.
Gittis’ findings will be put to the test in a human efficacy trial conducted by brain specialists at the Allegheny Health Network (AHN) in Pittsburgh.The controlled trial will consist of a group of people suffering from spontaneous Parkison’s disease, as well as a group of individuals without the disease. The individuals will not know whether they are receiving the DBS treatment or a simulated treatment. Researchers will observe all individuals for a year following the initial treatment to evaluate resolved motor issues and the quantity of continued spasms.
“Aryn Gittis continues to do spectacular research which is elucidating our understanding of basal ganglia pathology in movement disorders. We are excited that her research on burst stimulation shows a potential to improve upon DBS which is already a well-established and effective therapy for Parkinson’s disease,” said Nestor Tomycz, a neurological surgeon at AHN.
According to Donald Whiting, this approach may lay the foundation for innovative therapies. This is fantastic news, especially coming from the chief medical officer of AHN, who also happens to be among the nation’s foremost specialists on the use of DBS.
“Aryn is helping us highlight in the animal model things that are going to change the future of what we do for our patients. She’s actually helping evolve the care treatment of Parkinson’s patients for decades to come with her research,” Whiting said. “This work is really going to help design the future technology that we’re using in the brain and will help us to get better outcomes for these patients,” added Tomycz.
This study is published in Science.






-392x250.jpg)


Comments