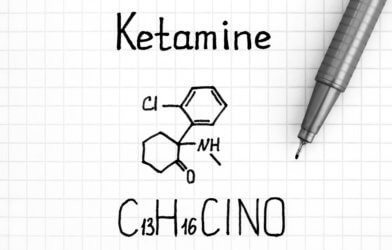
Ketamine is well-known as a popular party drug, especially in Western Europe. In 2019, the nasal spray esketamine, the first ketamine-based antidepressant, was received by the clinical industry with excitement for its potential in patients with mood disorders. The U.S. Food and Drug Administration still limits the spray’s use due to its ketamine content and safety implications. A new study conducted at the Weizmann Institute of Science in Rehovot, Israel, and at the Max Planck Institute of Psychiatry in Munich, Germany, in collaboration with the Helmholtz Zentrum, Munich has sought out to address this.
Previously, when scientists tried to clarify ketamine’s mechanism of action in other works, they examined its impact on genetic expression in brain tissue, but not in specific brain cells. This approach can miss valuable differences between differing cell types. Newer technologies have made it possible to assess expression with high resolution, which is why they’ve been used for this latest work.
To do this, the team mapped out expression in thousands of individual neurons in mice brains that had been given a ketamine dose. They focused on the ventral hippocampus, which is a brain region that in previous studies had been associated with the antidepressant effects of ketamine. After, the researchers identified a subpopulation of neurons with a unique genetic signature.
Ketamine had increased the neuronal expression of a gene called Kcnq2, which encodes a potassium channel. This means that a tunnel that opens up in the cell membrane enables the passage of potassium ions. Potassium channels play a central role in the life of neurons, maintaining their stability and preventing excessive energy bursts.
In a series of complex experiments, the researchers confirmed that ketamine exerts its lasting antidepressant effect by enhancing the Kcnq2 potassium channels in certain neurons. Following this, they then tested ketamine’s effects in combination with an epilepsy drug called retigabine, which is known to activate potassium channels in the brain. When given together, ketamine’s antidepressant effects were significantly heightened.
“A single dose of retigabine was enough to amplify and prolong ketamine’s antidepressant action in mice,” says Dr. Juan Pablo Lopez, in a statement. “Not only that, ketamine produced the same benefits when given in smaller doses than usual, which may help reduce its unwanted side effects.”
Depression affects millions of lives across the world, and despite years of research, there still remains much to learn about the mechanisms associated with it. By discovering ketamine’s mechanism of action, this work may provide a basis for expansion of ketamine-based drugs in safe doses. It’s the hope of the researchers that their work inspires scientists to look further into this in the future.
The study is published in the journal Neuron.