A new Phase 2 clinical trial by Green Valley Pharmaceuticals will soon test its seaweed-based oral compound oligomannate in early-stage Parkinson’s disease patients. This marks Green Valley’s next step after recently receiving U.S. Food and Drug Administration (FDA) approval on their investigational new drug (IND) application.
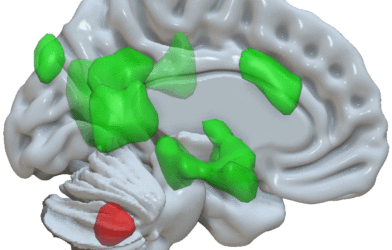
The rapid progression of Parkinson’s disease is thought to be associated with the aggregation of alpha-synuclein protein in the nerve cells, resulting in neuroinflammation. Recently, research has also found that gut microbiota may be involved in the occurrence and development of Parkinson’s disease. A similar connection between gut inflammation and neurodegenerative disorders has been found in recent Alzheimer’s disease research.
An oral compound called oligomannate, made from marine brown algae and said to help build a healthy gut microbiome, has been shown to be capable of targeting the connection between the central nervous system and gastrointestinal tract referred to as the “gut-brain axis.” Treatment with this drug works by retraining the microbiota in the gut and blocking imbalance of the byproducts of gut microbiota metabolism, leading to reduced inflammation.
Oligomannate treatment has been shown in preclinical studies by Green Valley researchers to assist in regulating gut microbiota. Preclinical studies carried out by Green Valley scientists have found that the therapy is able to help regulate abnormal balances in the gut microbiota and reduce aggregation of both gut and brain alpha-synuclein in Parkinson’s disease models.
In addition to promising reductions in protein aggregation, Oligomannate also proved capable of reducing inflammation in the brain, reducing both motor and non-motor Parkinson’s symptoms, and protecting the dopamine-producing neurons that are lost over the life of disease.
Phase 2 of the trial will take place over a 36-week period. The study will be placebo-controlled, happening across 30 clinics in North America and the Asia Pacific. The study will then enter a 36-week open-label extension period, where all researchers and participants are made aware of the drug treatment they are partaking in.
China has already approved Oligomannate for Alzheimer’s treatment and cognitive function. Green Valley hopes to achieve the same in the United States and has plans to include 300 early-stage Parkinson’s patients in their study to ensure the treatment is safe and effective.
In China, trials of oligomannate reached a third phase in which 818 patients with mild to moderate Alzheimer’s were treated with the drug and found to have had positive impacts on cognitive function without significant detrimental side-effects.
“We are deep diving into the gut-brain axis study with a holistic view of treatment. Those insights are expected to become a major driving force to help us address unmet medical needs,” says Green Valley’s website. It is possible, they hope, “to uncover a new angle to interpret the pathogenesis [origination and development] of chronic and complex diseases.”
The news covered in this article was originally published in Parkinson’s News Today.
Article written by Anna Landry