Researchers from the Department of Biomedicine at Norway’s University of Bergen (UiB) believe drugs could be found to catch Parkinson’s disease at an earlier phase, preventing it from progressing further. They believe the “happiness” hormone dopamine can play a key role in achieving the feat.
Experts have long known that high levels of the hormone dopamine have shown positive effects on the overall emotional well-being of a person. With billions of nerve cells that use neurotransmitters to communicate, dopamine has many functions, including the impact it has on daily activities such as concentration, motor control and learning and attention. The hormone has been tied to generating feelings of happiness and an increased sense of pleasure and reward.
With patients who have Parkinson’s disease, the brain cells that produce and secrete dopamine die. In most cases, by the time a patient is diagnosed with Parkinson’s, more than half of the cells are gone and significant damage has taken place. Thus, making it difficult to diagnose at an earlier stage in the disease process.
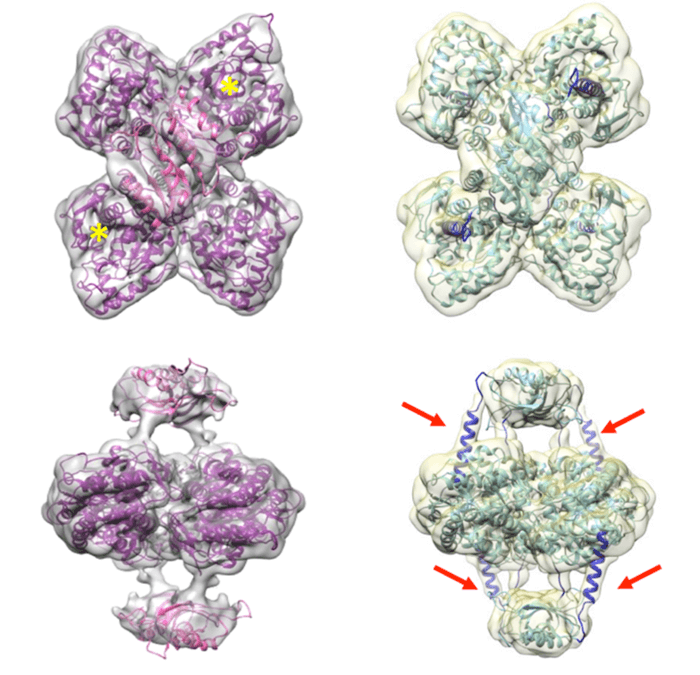
When exploring the process of how dopamine regulates its own production, enzymes are responsible for instigating chemical reactions in the body. A specific enzyme called tyrosine hydroxylase (TH) has been found in large amounts inside dopamine-producing cells. These three-dimensional structures of enzymes illustrate cellular processes at the atomic level. When errors are found resulting in mutations, the mutations can cause deficiency. This concept is also seen with TH deficiencies. These TH deficiencies are known to cause neurological problems, such as Parkinson’s.
In collaboration with the Neuro-SysMed Center, the Bergen research team is exploring how proteins operate at a structural level. By learning more about how the mutations cause defects in protein function, they hope to better understand how defects can be repaired.
“Such 3D structures can tell us how the cellular processes take place at the atomic level and thus also how we can find targeted treatment to correct errors in enzymes, which, for example, occur by disease mutations. For TH, such mutations cause TH deficiency, a neurological disease classified as a subgroup of parkinsonism”, says Professor Aurora Martinez, with UiB’s Department of Biomedicine, in a statement.
It’s widely known that dopamine has the ability to regulate its own production and bind to the TH enzyme, causing it to inactivate. This process of regulation is known as negative feedback and has the ability to be turned off when adequate levels of dopamine have been produced.
Likewise, when dopamine levels drop, the enzymes may be activated in the cell again, causing TH to be modified. This modification causes dopamine to be released and the enzyme to be reactivated. It is this process of dopamine regulation researchers are able to observe and better understand.
Cryo Electron Microscopy has provided high resolution technology for researchers to explore what TH looks like and observe changes that take place when it binds to dopamine. This advanced technology has made it possible to observe three-dimensional structure proteins in great detail. Researchers at the Department of Biomedicine and Helse Bargen and the Centro Nacional de Biotecnología in Madrid, led by Professor José Maria Valpuesta, collaborated using this technology.
As a result of this study, new medicines may be created for neurodegenerative and neuropsychiatric disease as knowledge becomes increasingly advanced about the TH enzyme and its binding processes to the hormone dopamine.
The findings from this study were published in the journal Nature Communications.
Written by Elizabeth Bartell