Within the science field, researchers have tried to understand the relationship between obesity and brain signaling. It’s been previously understood that the hormones in the blood were responsible for fat tissues passing information on stress and metabolism to the brain. Now, scientists from the Scripps Research Institute have identified specific sensory neurons that carry memos from the fat tissue to the brain.
“The discovery of these neurons suggests for the first time that your brain is actively surveying your fat, rather than just passively receiving messages about it,” says co-senior author Li Ye, PhD, the Abide-Vividion Chair in Chemistry and Chemical Biology and an associate professor of neuroscience at Scripps Research, in a statement. “The implications of this finding are profound.”
So far, researchers have agreed that nerves extend into adipose tissue, but they didn’t actually think that sensory neurons played an active role in carrying information to the brain. Most have hypothesized that the nerves within fat tissues belonged mainly to the sympathetic nervous system, the network in charge of our fight-or-flight response. It’s been a tedious task, however, to clearly identify the different types and functions of these sensory neurons because the methods utilized for examining neurons do not transfer well for use in fat tissue since the nerves are harder to see and reach.
In this work, the research team developed ways to work through it, successfully generating two new methods that help with this issue. First, they took an imaging approach called HYBRiD, which is a technology able to turn mouse tissue transparent. This allows the researchers to more clearly track how the neurons are moving into the tissues. They found that nearly half of the neurons didn’t even connect to the sympathetic nervous system; rather, they did to the dorsal root ganglia, which is the part of the brain where all sensory neurons come from.
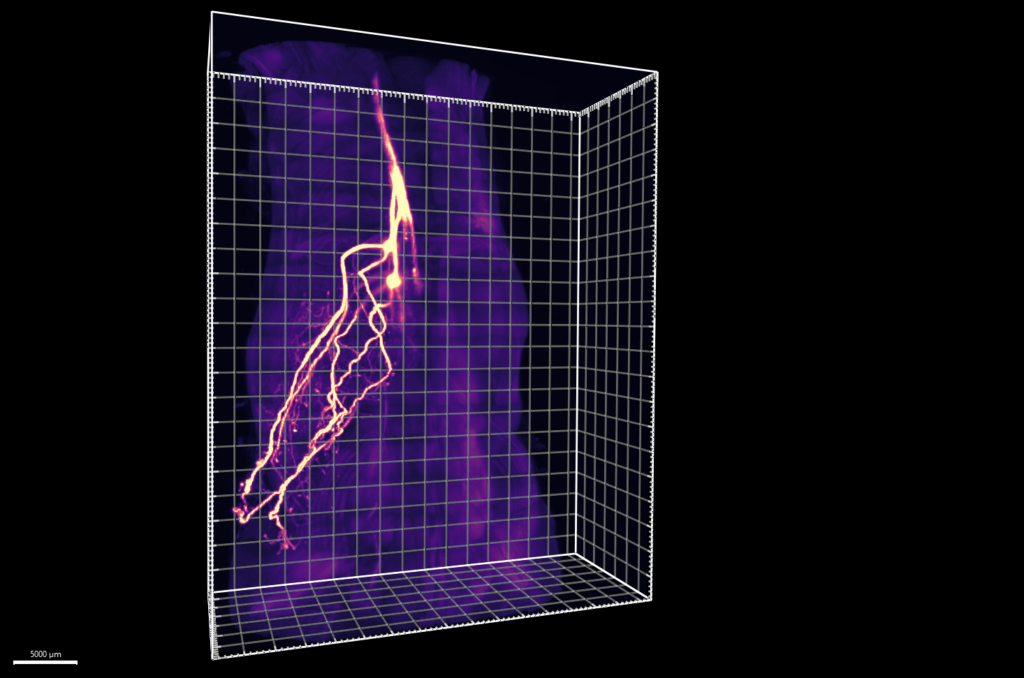
To gain a clearer understanding on what the neurons do in fat tissue, the team created an additional technique called ROOT, which stands for “retrograde vector optimized for organ tracing.” This allowed for them to selectively destroy small subsets of sensory neurons inside the tissues using a targeted virus, and then examine the subsequent happenings.
They ultimately concluded that when the brain doesn’t receive sensory messaging from fat tissue, programs triggered by the sympathetic nervous system become overactive. Since this area converts white fat to brown fat, this results in an abnormally large fat pad with lots of brown fat. As brown fat is related to temperature regulation, this mechanism was backed in animal models with blocked sensory neurons because they consequently had high levels of sympathetic signals and raised body temperatures.
“This tells us that there’s not just a one-size-fits-all instruction that brain sends adipose tissue,” explains Li. “It’s more nuanced than that; these two types of neurons are acting like a gas pedal and a brake for burning fat.”
Although the team doesn’t exactly understand what the messages say between fat tissue and the brain, knowing that there is any connection at all is valuable for maintaining optimal fat. They plan to conduct further work to understand exactly what is being sensed, and if any similar mechanisms are seen in other areas of the body.
This study is published in the journal Nature.