Alzheimer’s disease has been reversed in mice after scientists boosted the formation of new brain cells. The breakthrough could lead to new treatments for the incurable, memory-robbing condition.
The study offers hope of a viable strategy. Current drugs target just the symptoms, and not the cause. Scientists now say a gene therapy they developed fueled neurons in the hippocampus, a region vital for learning and remembering.
“Taken together, our results suggest augmenting neurogenesis may be of therapeutic value,” says lead author Orly Lazarov, a professor from the Department of Anatomy and Cell Biology in the University of Illinois Chicago College of Medicine, in a statement.
Brain cells send electric signals. We keep making them throughout our lives. They are produced by neural stem cells. But numbers tail off as we age and fall dramatically in Alzheimer’s. Evidence is improving neurogenesis holds the key to curing dementia.
“However, the role of newly formed neurons in memory formation, and whether defects in neurogenesis contribute to the cognitive impairments associated with Alzheimer’s, is unclear,” says Lazarov.
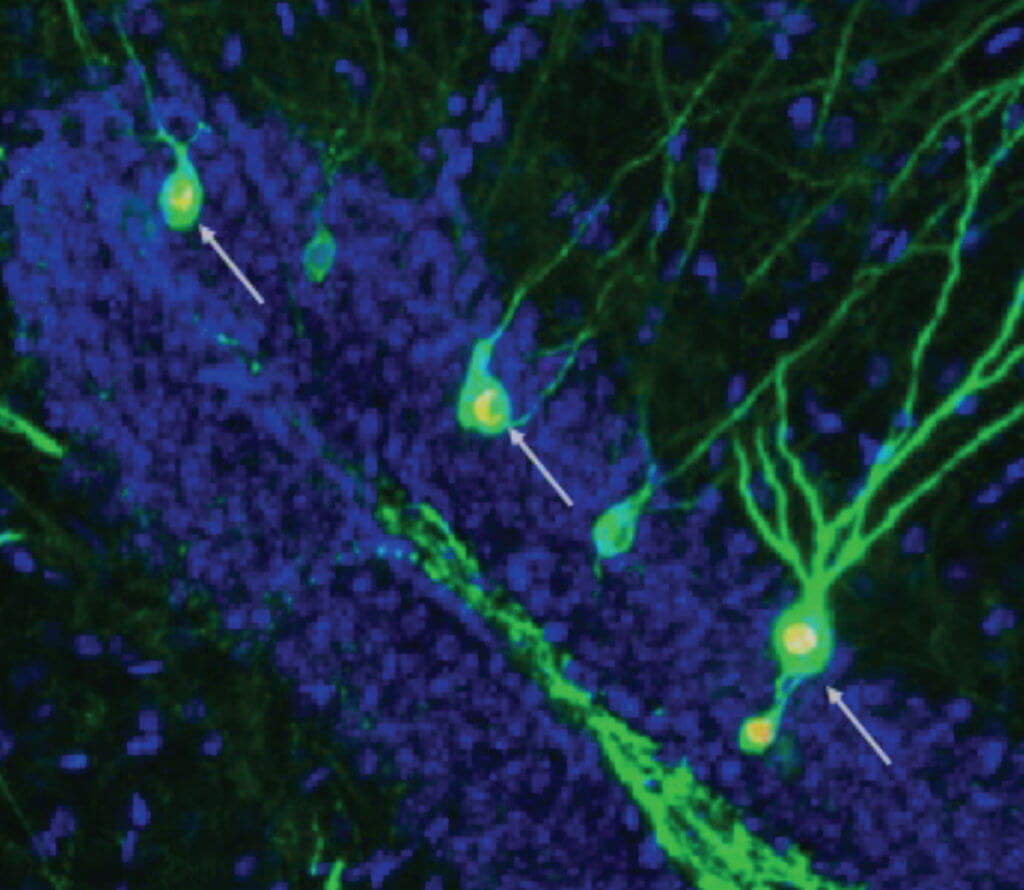
Experiments have shown the process is impaired in patients and mice with mutations linked to Alzheimer’s, particularly in the hippocampus. The hippocampus is the region of gray matter you need, for instance, to remember where you parked your car. Study authors found increasing production of neurons rescued the lab rodent’s defects. They were incorporated into memory circuits, restoring normal function.
In the study, stem cell survival was enhanced by deleting a gene called Bax, leading to the maturation of more neurons. Afterwards, the animals regained the animals’ regained their spatial recognition and contextual memory skills. Tests included finding their way around a maze.
Scans of healthy mice showed the circuits involved in storing memories include many newly formed neurons alongside older ones. Neurons were fluorescently labelled, lighting up as they were activated during acquisition and retrieval. The memory-stowing loops of mice with Alzheimer’s contained fewer. But integration of newly formed brain cells was restored when neurogenesis was increased.
Further analyses revealed there was also a rise in the number of tiny protrusions called dendritic spines. They connect neurons and are critical for memory formation.
When the researchers specifically inactivated the new neurons, mice with dementia lost any improvement in memory, confirming the results.
“Our study is the first to show impairments in hippocampal neurogenesis play a role in the memory deficits associated with Alzheimer’s by decreasing the availability of immature neurons for memory formation,” says Lazarov.
The number of dementia cases worldwide will triple to 150 million by 2050. The study is published in the Journal of Experimental Medicine (JEM).
Report by Mark Waghorn, South West News Service